Gravity separation works well with lodes or veins, but there are few of those types of deposits to be found. 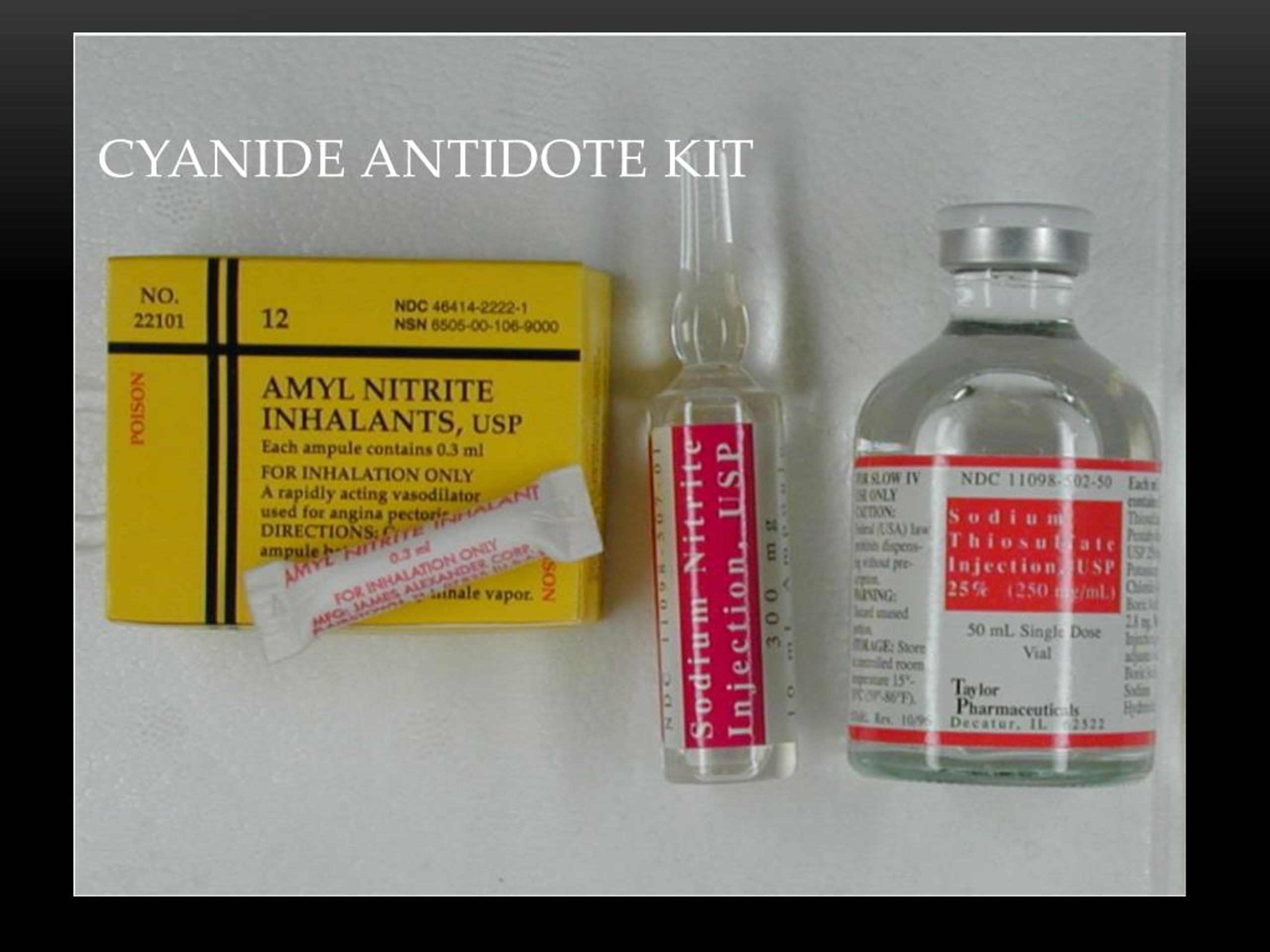
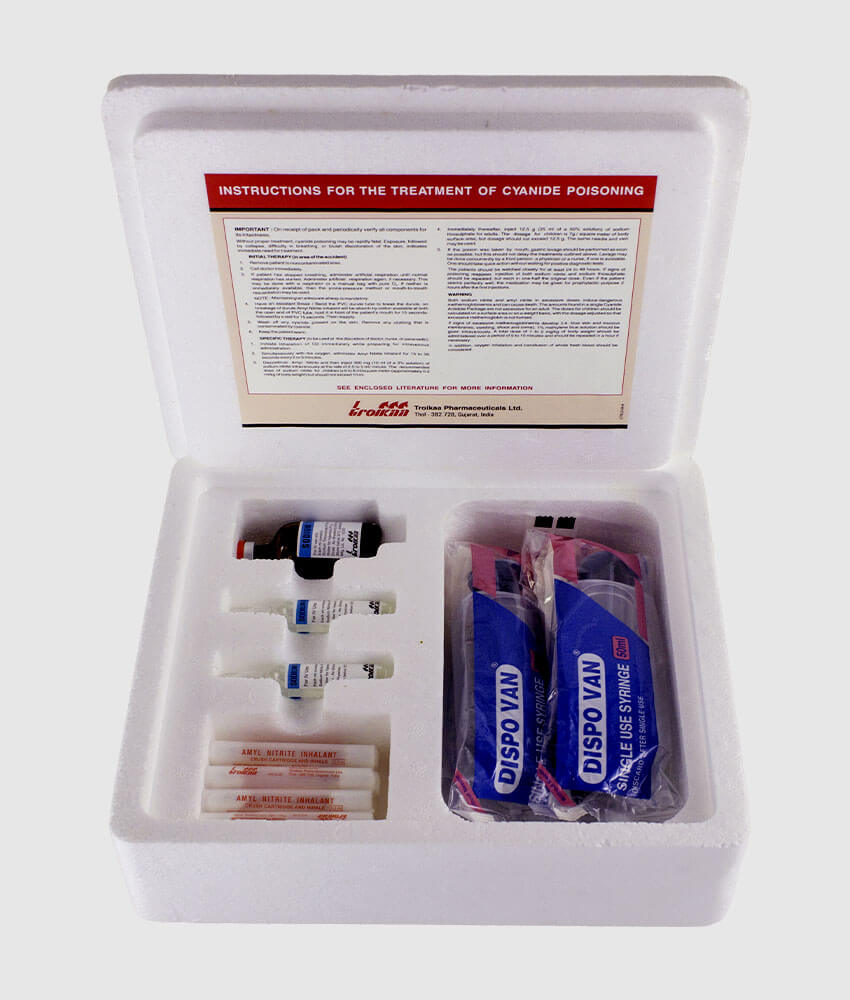
In other words, banning the use of cyanide in effect bans the extraction and production of gold. It can easily be separated from gold for collection. Any remaining cyanide decomposes naturally as sunlight breaks the compound into carbon and nitrogen.Ĭyanide is a naturally occurring compound that bonds with gold, allowing it to be brought into solution. Some operations may also use water treatment facilities to treat and neutralize effluent discharge. The detoxification and neutralization process converts cyanide into a less toxic cyanate, before combining it with the mine tailings. When the mine is closed, there is a short- and long-term cleanup plan to detoxify the solution so that no residual cyanide remains to harm the environment. During operations, the cyanide solution is collected and processed. Mines contain the cyanide solution by placing multiple “impermeable” liners at the base of heap leaching stockpiles and along the bottom of leachate collection ponds. Heap leaching is by far the widest application and the most controversial. States that currently use cyanide leaching to recover metals from ore are: Arizona, California, Colorado, Idaho, Montana, Nevada, Utah, South Carolina, South Dakota and Washington.Ĭyanide solution can be used for both heap leaching and vat leaching applications. The State of Nevada alone annually contributes approximately 75% to national U.S. gold mining industry, accounting for the most exploration and mining activity on federal lands. With more than 150 gold mines, Nevada is the hub of the U.S. The remaining 94% of hydrogen cyanide is used to produce a wide variety of products, such as adhesives, computer electronics, fire retardants and nylon. Of the 1.1 million tons of hydrogen cyanide (HCN) produced annually, only 6% is converted into sodium cyanide for use in the mining industry. 
More than 90% of all gold extracted worldwide relies upon the use of cyanide. While there have been notable past problems with cyanide, for the most part its use by the mining industry has not resulted in environmental catastrophe. These groups often rally the support of local citizens (voters) by conjuring the memories of those disasters. Nonetheless, despite a voluntary policing effort by the industry, and the adoption of strict regulations by governments where the mines are located, certain environmental groups have attacked the use of cyanide. In part this basically good record is due to an evolving regulatory landscape, and a realization by the mining industry that cyanide spills are to be avoided. However, worldwide, the mining industry has shown it can work with cyanide responsibly. The chemical compound has on occasion been mishandled, and in some locations releases have created environmental problems. The use of low-cost cyanide heap leaching techniques to recover metal from ores has allowed miners to profitably extract gold deposits. Memories of past disasters reinforce present-day fears 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
